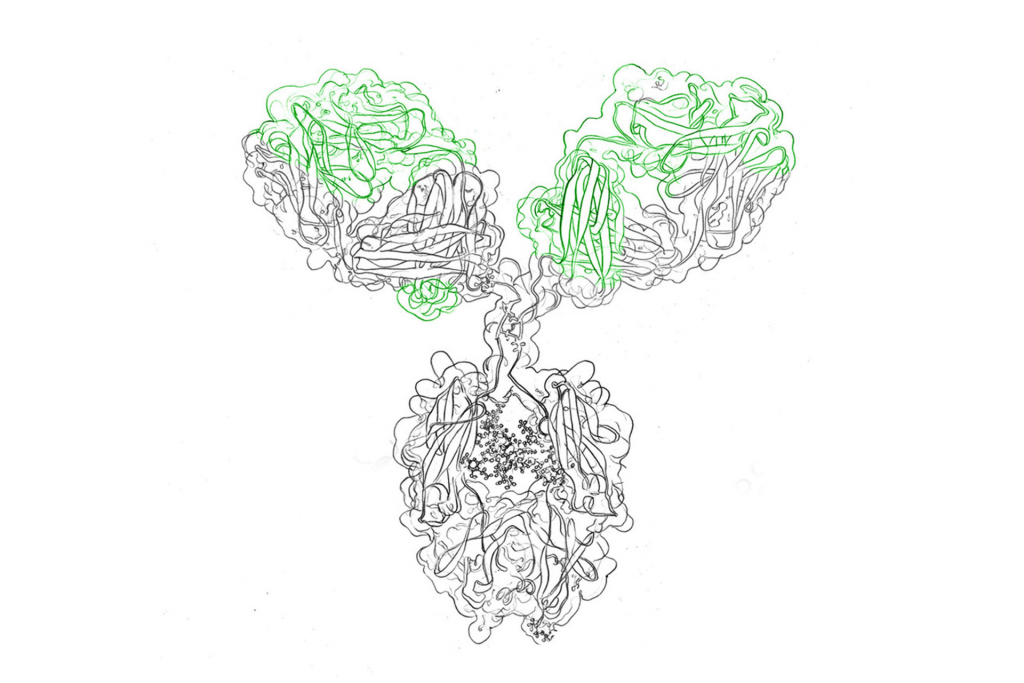
Therapeutic antibodies with mutated Fc regions demonstrate reduced Fc-effector functions (such as cytokine release) in vivo. This is essential for therapeutic applications where activation of the host’s immune system would have negative effects, such as when an antibody is used to neutralize a binding interaction.
Many mutations exist which silence the Fc domain, but all previously described version have some background activity and intellectual property limitations.
evitria holds a licensing partnership to offer antibodies with truly silent Fc domains, using mAbsolve’s STR technology. These antibodies display zero activity above background noise.
Antibody production runs
Lead time
Projects completed on time
STR is a novel combination of three mutations in the CH2 domain of the Fc. STR-modified antibodies show no binding or activity above background noise across the Fc gamma receptors, far surpassing all previously described modifications.
STR is readily available to license, and evitria is partnered with mAbsolve to offer truly silent Fc domains for preclinical use.
Sourcing STR-silenced antibodies through evitria enables our clients to benefit by gaining:
From sequence to antibody in 5 weeks
Project start
0-1 business days

Pilot study
(from electronic sequence to quantified pilot supernatant)
15-20 business days
Larger-scale expression
5-10 business days

Purification
2-3 business days

Final analytics
2-3 business days
Shipment
1-2 business days
How to order with evitria
You tell us what you need and when
Optional: We discuss your project and how we might be able to assist in a personal conversation
You receive our proposal within one working day
You review and approve our proposal
Your project begins within one working day
For your projects in Medicine or Research, both quality and time are of essence. As an antibody expression service provider, we help our customers out with a fast delivery of high-qualitative antibodies produced with recombinant technology.
At evitria, we provide not only Fc-silenced antibodies, but also
In addition to this, our expert team conducts a careful sequence and project review, so that we can provide you with high-quality antibodies for your project – from sequence to antibody in 5 weeks.
After your approval, we begin with your project within one business day!