Antibody-dependent cellular cytotoxicity (alternatively antibody-dependent cell-mediated cytotoxicity or ADCC) is a key process of the immune system’s arsenal to ward off cancerous cells and infections.
When a pathogenic cell is recognized and covered by specific antibodies, it becomes a target for effector cells. In the course of a distinctive immune response, natural killer cells will capture target cells, inject them with cytotoxic factors and ultimately lyse it to harmless fragments.
Domestication of this mechanism is of high interest in the fields of medicine, biotechnology and life-sciences, enabling the development of profitable products. Read more about the ADCC process or ADCC antibodies in this article.
In immunology, ADCC stands for Antibody-Dependent Cellular Cytotoxicity and is a mechanism of the humoral immune system against pathogenic cells and microbes.
Subscribe to our Newsletter
Get all the latest updates, and learn about our advancements in antibody production.
Subscribe now
The definition of ADCC is the process of antibodies recognizing and binding to pathogens, essentially covering their surface to highlight them to specialized blood cells termed natural killer cells (NK cells). NK cells in turn kill those intruders.1
ADCC enhanced antibodies are a special class of antibodies that lead to stronger ADCC responses of the immune system.
Compared to normal antibodies, ADCC enhanced antibodies have a modified glycosylation pattern that lacks fucose. This is the signal for NK cells to step up their cytotoxic activity.
The intentional manipulation of antibodies ex vivo to have them lack fucose is termed afucosylation. Such afucosylated antibodies are typically manufactured by recombinant technology in mammalian cell lines.
They hold high promise as novel therapeutics against cancer and infectious diseases and hence are in high demand in discovery and development research.
Further readings:
Antibody-dependent cellular cytotoxicity, also termed antibody dependent cell mediated cytotoxicity (ADCC) – is an important weapon in the immune system’s arsenal to fight cancerous cell types and infected cells.
The process has been described by US and Korean scientists in “Antibody Fc – Linking Adaptive and Innate Immunity in 2014”.2
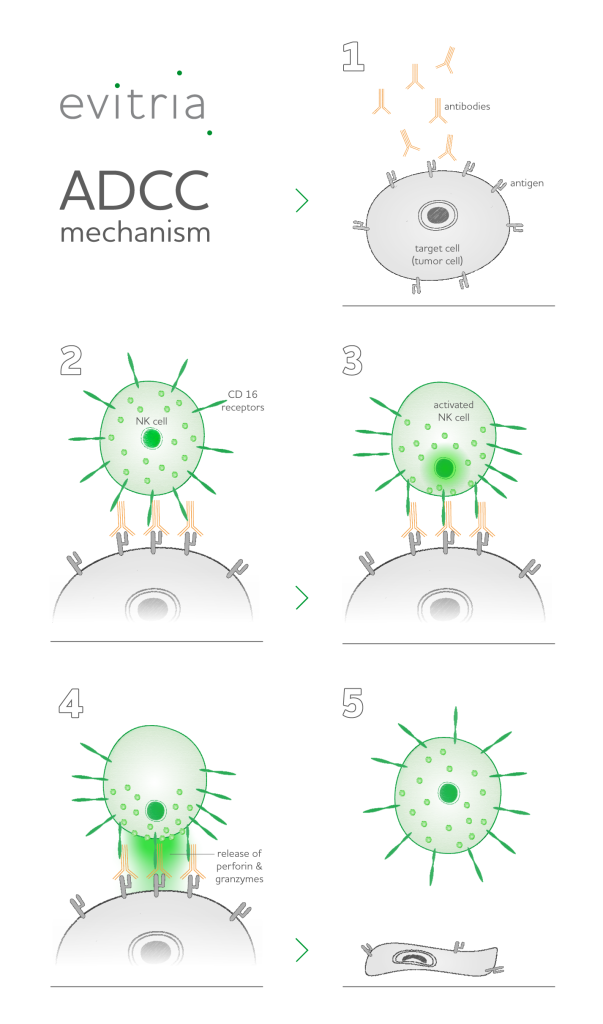
The mechanism of mAB-mediated ADCC relies on immunoglobulins (IgG antibodies) that bind to the target cell’s surface with their highly specific antigen binding sites. The other end of the antibodies contains a glycosylated region that represents binding sites for effector cells like peripheral blood mononuclear cells, mostly natural killer cells.
A class of Fc receptors on the surface of NK cells, termed CD16 receptors, binds to these antibodies, which results in cross-links between CD16 receptors. Subsequently, a cascade of signal transduction pathways is put in motion to activate NK cells.
Upon activation, NK cells release granules filled with cytotoxic factors, termed granzymes and perforin onto the target cell. Perforin inserts into the target cell’s membrane and forms pores through which granzymes can enter the inside of the target cell.
Granzymes are protease proteins that attack target cells from the inside, leading to cell death. Ultimately, the NK cell lyses the pathogenic target cell into harmless fragments that are digested and recycled by the body.
Speaking of Antibody dependent cell mediated cytotoxicity, it is important to highlight natural killer cells as very potent key players in the body’s fight against cancerous cells and infections.
One of their virtues is that they are less discriminate in antibody-dependent cellular cytotoxicity (ADCC) than the immune complement system, since they can be activated by antibody binding, in contrast to specific antigen binding.
Hence, their therapeutic use by means of ADCC holds high promise in broad fields of medicine and medical research applications.
Numerous monoclonal therapeutic antibodies have been shown to mediate their effects partially or predominantly via NK cell activation through antibody-dependent cell cytotoxicity (ADCC):
In recent years, novel therapeutic recombinant antibodies have been engineered to exhibit enhanced ADCC activity.
ADCC enhanced antibodies are recombinant antibodies that are optimized to exhibit increased antibody-dependent cellular cytoxicity.
Antibody-dependent cell-mediated cytotoxicity is mediated through binding of NK cell’s CD16 receptors to the Fc region of antibodies.
It has been shown in vitro that lack of fucose in the Fc region leads to stronger binding of CD16 receptors, hence shutting out non-specific competition with other antibodies and increasing the ADCC response.
Intentional production of recombinant antibodies that lack fucose (afucosylation) for discovery research applications has delivered evidence that strengthens these expectations.
These findings are highly promising for the development of better treatments for cancer and infections. ADCC enhanced antibodies show two key benefits over conventional therapeutic antibodies:
In the particular case of ADCC (antibody-dependent cellular cytotoxicity) enhanced antibodies, the binding properties are complemented with a distinct additional mechanism of action to the usual IgG1 antibody response: the lack of fucose in the antibody Fc region enhances the binding with Fc receptors or FCRs (Fc gamma / FcγR, namely FcγRiii and FcγRiiia), resulting in enhanced effector functions, especially recruitment of effector cells such as natural killer cells (NK cells) with their perforin enzymes towards target cells.
The overall result is a very aggressive immunotherapy that draws immune cells such as lymphocytes (T cells, B cells), monocytes and macrophages towards tumor cells, virus particles, microbes and other pathogens. The ADCC response correlates with increased cytokines, lysis, phagocytosis, granzyme release, apoptosis, all leveraging immune cell activation against tumorigenic and infected cells.
The popularity of ADCC enhanced antibodies is consistently growing. Since any type of genetically modified and cultivable antibody is of interest for players in the biopharmaceutical and life-science industries.
Thanks to a number of beneficial properties, they can be used in a number of medications and treatments targeting autoimmune diseases, cancer and viral diseases such as COVID-19. The two major benefits are:
The production of afucosylated monoclonal antibodies relies on the attachment of fucose-lacking oligosaccharides to the antibodies in host cells.
The most effective way to achieve this, is the use of high yielding cell lines that are genetically engineered to have enzymes that block the incorporation of fucose into expressed antibodies. One embodiment of this approach, namely the use of highly advantageous CHO cell lines concurrent with ProBioGen GlymaxX® technology, is used by evitria with large success.
ADCC enhancement technology is expected to play a key role in the development of therapeutic antibodies with improved clinical efficacy. A number of preclinical and clinical trials regarding the application of non-fucosylated antibody therapeutics are currently underway.
But why are ADCC antibodies seeing such a steady rise in popularity? Well, they exhibit a number of significant benefits, the three major ones being:
As several studies have shown, enhancing the potency of ADCC could be an effective way of combating influenza virus infection. But it doesn’t stop here: ADCC may be effective in controlling other infectious diseases – like the Corona virus currently causing so much havoc – too.
ADCC has been shown to provide cross-strain protection and to assist in viral clearance, making it an attractive target for “universal” vaccine designs. ADCC-mediating antibodies also target other proteins on the virus surface, making it a potentially important mechanism for protective immunity in herpes simplex virus infections.
Researchers in immunology are able to design specific antibodies in formats that target a certain antigen, such as the one found on cancer cell surfaces before then multiplying said antibody in the lab. The result are monoclonal antibodies (mAbs) that are used to treat many diseases (antibody expression COVID-19), including some types of cancer.
ADCC enhancement is important to push the efficacy of cancer antibodies as the artificially decreased fucose content that leads to ADCC also results in an elevated killing activity against tumorigenic and infected cells, making them more powerful and efficient.
So it is hardly surprising that the popularity of ADCC enhanced antibodies to be utilized in the development of next-generation therapeutic antibodies is constantly growing.
The constantly growing demand is just one of the reasons why antibody production and design play a crucial role in today’s life-science industry, be it to increase activity as well as functionality, reduce immunogenicity or extend the half-life.
The scientists at evitria are specializing in the recombinant antibody expression service including afucosylated antibodies for ADCC enhancement. One of the most recent additions to the company’s services is the state-of-the-art GlymaxX® technology, licensed from ProBioGen, used for the generation of afucosylated antibodies by means of transient expression in CHO cells.
The company holds an exclusive licensing partnership to offer afucosylation via ProBioGen’s GlymaxX® technology for your transient antibody expression needs. This technology facilitates the expression of both native and afucosylated variants.
The technology can be applied to your standard host cell line. Alternatively, we can offer our own optimized GlymaxX host cell line, or even re-engineer existing producer clones. GlymaxX cell lines are stable and compatible with highest titer production processes.
Determined by the effector cells concerned with ADCC, IgG, but also IgE antibodies or other non-neutralizing antibodies can be involved.
ADCC immunology – ADCC (or antibody-dependent cellular cytotoxicity) is an essential immunological process; antibodies bind to the surface of specific target cells to then be recognizable for effector cells – NK cells, macrophages, neutrophils or eosinophils –, which then aim to destroy the targeted cells.
The main function of ADCC is to protect an organism from (potentially) harmful cells; target cells are covered by antibodies to then be recognizable for effector cells. These, consequently, aim to destroy the target cells.
ADCC commences as soon as effector cells recognize a target cell to which specific antibodies are bound. They then seek to attack and destroy the targeted cells. Consequently, antibodies play an essential role in this process, not only able to initiate antibody-dependent cellular viral inhibition, but also ADCC.
Antibody-dependent cellular cytotoxicity (in short ADCC) is a central mechanism of the immunity system: Antibodies are bound to specific target cells, which consequently can be detected and eliminated by effector cells.
ADCC is an essential component in several immunological processes concerned with the protection of an organism from (potentially) harmful cells. Additionally, the process is of great interest for researchers on the attempt to develop groundbreaking new therapeutic approaches, e.g. in tumor therapy, which was shown in mouse models.
Antibody glycoengineering is a process that modifies the sugar (glycan) structures on antibodies to enhance their biological functions. This can involve changing the glycans to promote ADCC (Antibody-Dependent Cellular Cytotoxicity), which boosts the immune system’s ability to target and destroy infected or cancerous cells. One specific glycoengineering approach is to create afucosylated antibodies, which have reduced fucose sugars on their glycans, resulting in increased ADCC activity.
ADCC is caused by the recognition of harmful cells, which then specific antigens bind to in order to make the target cells visible for effector cells, which then aim to destroy them.