From sequence to antibody in 5 weeks – with the highest quality standards
Low levels of endotoxin <1EU/mg
As the number of new biopharmaceuticals entering the market is growing, the importance of Contract Research Organizations, or CROs in antibody production is increasing simultaneously.
The production of monoclonal and recombinant antibodies is a laborious multi-step process that requires the right equipment and expertise. For this reason, many pharma companies rely on Contract Drug Manufacturing Organizations, or CDMOs, in antibody production because they do not have the required infrastructure at their disposal. After an antibody drug has been developed by the CDMO, a CRO is consulted for drug testing and the conduction of clinical trials.
In this article, we will explain the purpose of CROs, their importance for the industry and, especially, for the field of monoclonal antibody production.
A CRO (Contract Research Organization) is a company that offers research services to other companies which are active in different sectors of life sciences, including biotechnology, healthcare or biopharma.
Subscribe to our Newsletter
Get all the latest updates, and learn about our advancements in antibody production.
Subscribe now
Especially in biopharma, companies often outsource the production and the development of specific antibodies as well as antibody discovery to CROs because high quality custom antibodies are the baseline to every successful antibody drug.
Due to the developments in monoclonal antibody (mAb) production on an industrial level, it became possible to generate highly effective antibody drugs against complicated diseases. In contrast to monoclonal antibodies, polyclonal antibodies have a lower specificity and target multiple epitopes of an antigen, making them more susceptible to cross reactivity, but also more complicated to produce.
Monoclonal antibodies or recombinant proteins like small peptides are mainly produced in mammalian hybridoma cell lines, using in vitro hybridoma technology. Mammalian cell cultures typically used for this process range from HEK 293 cells to CHO cells, because they show lower toxicity levels and are able to produce IgGs (immunoglobulin G) that are similar to human antibodies.1
Further, purification and testing with Western blots and Enzyme-linked immunosorbent assay or ELISA in order to test receptor binding efficiency are necessary steps in terms of quality control.
Depending on their specialization, CROs provide preclinical research, clinical trials, biostatistics and a variety of other research-related, but also marketing-related services to other companies.
The generating and characterization of an antibody are often handed over to CROs that specialize in antibody development because they have access to the required technology and equipment. While polyclonal antibodies are often produced in vivo, injecting a living animal and triggering its immunization response, the production of monoclonal antibodies requires hybridoma development in fusion with myeloma cell lines, phage display or B-cell cloning. CROs offer phage display libraries and have the required bioreactors for protein production at hand to reach best results.
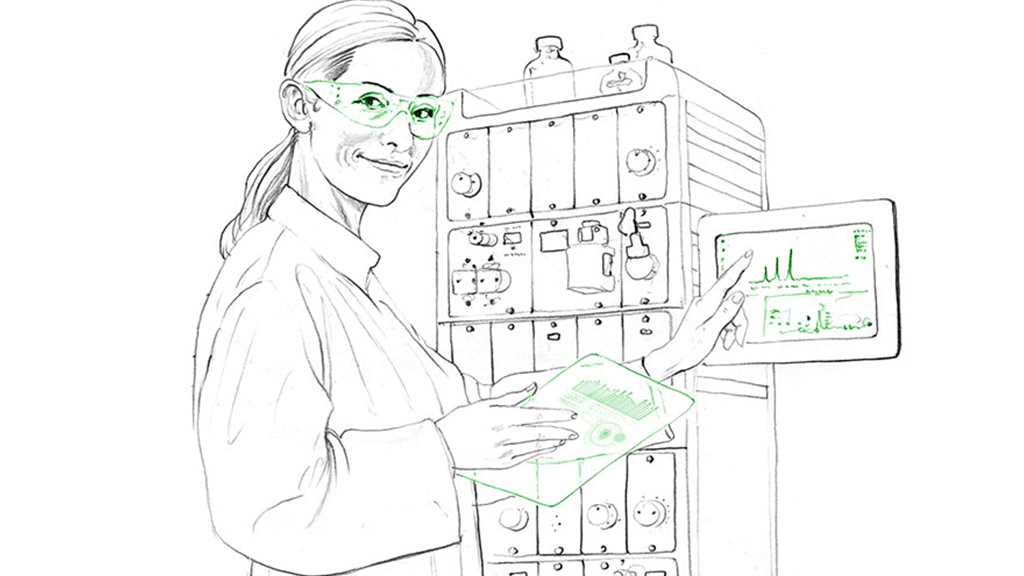
Especially for antibody-drug conjugations, a synthesis between an antibody and a drug with high cytotoxicity often used in the treatment of tumors or malignant ascites, it is important that the antibody is able to recognize the specific protein antigen to increase the immunogenicity of a drug. This is why amino acids are modified and tested with certain selective reagents by different CROs. Optimization and quality control of monoclonal antibodies has a high impact on the efficiency of a drug.
As the production of monoclonal antibodies on a large scale poses a major challenge, CROs with newest equipment like 96-well plates, shaker flasks and bench-scale bioreactors are able to perform high-throughput screening of antibodies and high titer masses. They can perform process characterization, scale-up and antibody validation for drug development, as well as antibodies with fluorescent markers which are used for diagnostic purposes and clinical assays.
Read more: Antibody production services – a comparison
Low levels of endotoxin <1EU/mg
The size of the CRO market makes up USD 56.7 billion and has an estimated growth expectancy of 6,9% in 2032. It is estimated that the market value will have reached USD 108 billion in the next ten years, as the demand for CROs in pharma is ever-growing. Over the last decade, the growth of CROs has shaped the biopharmaceutical industry. CROs made it possible for smaller companies to join the market by supplying them with the right equipment and expertise.
In most cases, working with a CRO proves to be beneficial for the partner company. However, there can be cases where the partnership is not successful because the CRO does not live up to the expectations. As outsourcing antibody production also means less control over the overall process, it is important to only enter a partnership with high-quality CROs. Nevertheless, the Pros and Cons of CROs seem to be heavier on the Pro-side if a trustworthy and communicative CRO is selected by the other party.
These are five advantages of CROs in monoclonal antibody production:
Read more: How to choose a partner for custom antibody production
evitria stands for customized recombinant antibody production services. With a broad range of services, high-end equipment and expertise, evitria provides support for your individual high-quality antibody project. Whether you are looking for recombinant antibody production on large-scale or a smaller-scale production for research purposes – evitria has got you covered.
With more than a decade of experience in the field, we can provide you with high quality recombinant antibodies in under five weeks from sequence to antibody.